Details
Currently, embryos are frozen at the pronuclear stage (2PN – one day after egg collection), the 2nd or 3rd day after egg collection (cleavage stage) or the 5th day after egg collection at the blastocyst stage.
 It is recommended that only good quality embryos should be frozen since only these embryos survive thawing (internationally around 80%) and give realistic possibilities of achieving a pregnancy. In any case, the couple should first discuss with their doctor upon the strategy and appropriate timing of cryopreservation and then make a decision in common.
It is recommended that only good quality embryos should be frozen since only these embryos survive thawing (internationally around 80%) and give realistic possibilities of achieving a pregnancy. In any case, the couple should first discuss with their doctor upon the strategy and appropriate timing of cryopreservation and then make a decision in common.
The primary goal in cryopreserving surplus embryos is to avoid the procedures of controlled ovarian stimulation and egg retrieval, in case that the “fresh” cycle fails to achieve a pregnancy. The woman can proceed to a frozen embryo transfer with optimal clinical outcomes.
Pregnancy rates generated by the transfer of cryopreserved embryos are slightly lower than those of “fresh cycles”. Furthermore, even if a pregnancy is achieved during a “fresh cycle”, cryopreserved embryos can be used when the couple decides to have a second child, eliminating the need for a new stimulated cycle.
Human embryos contain large amounts of water and are very sensitive during cryopreservation. In practice, the cooling procedure is always the same and involves placing embryos in specialized cryoprotectants, small neutral solutes, and final preservation under liquid nitrogen in cryostraws. Each cryostraw is sealed and labeled with the patient’s name.
Methods of cryopreservation
There are two methods for cryopreservation:
A. Controlled slow freezing and
B. Deep supercooling (vitrification).
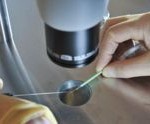 In controlled slow freezing, the cooling rate of oocytes/embryos is 0,3°C/min, whereas in vitrification the cooling rate is over 20.000°C/min. Even though the efficacy of vitrification was known many years before, its application to human oocytes/embryos became wide after 2010 as an alternative method offering several advantages. Vitrification prevents the damage caused by intracellular ice formation and therefore the survival rate after thawing is increased. Moreover, vitrification is an effective tool for oocyte freezing, offering high rates of survival and fertilization after thawing. These rates are comparable to using fresh oocytes.
In controlled slow freezing, the cooling rate of oocytes/embryos is 0,3°C/min, whereas in vitrification the cooling rate is over 20.000°C/min. Even though the efficacy of vitrification was known many years before, its application to human oocytes/embryos became wide after 2010 as an alternative method offering several advantages. Vitrification prevents the damage caused by intracellular ice formation and therefore the survival rate after thawing is increased. Moreover, vitrification is an effective tool for oocyte freezing, offering high rates of survival and fertilization after thawing. These rates are comparable to using fresh oocytes.
Cryopreservation has no apparent negative impact on the perinatal outcome according to literature. Available data indicate that children born after freeze-thaw procedures are healthy, even if embryos have been cryopreserved for many years.










